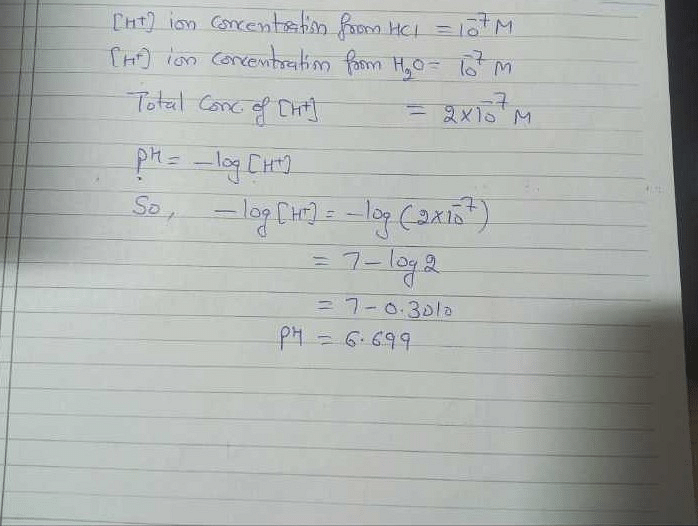31 0.1 ml 0.001 M hcl solution is diluted with water to make 10 litres .calculate PH of the dilute solution.

Assertion : `pH` of `10^(-7) M HCl` is less than `7` at `25^(@)C`. Reason : At very low concentratio - YouTube
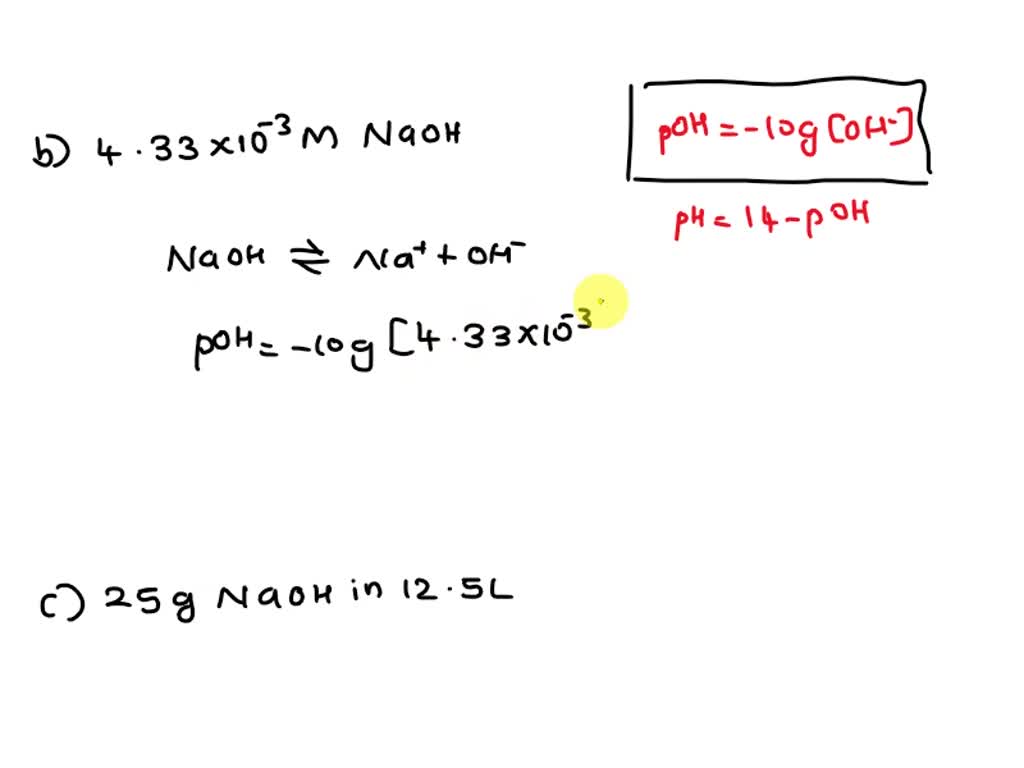
SOLVED: 7) What are the pH values of the following solutions? a) A 1.45 x 10-5 M HCl solution. b) A 4.33 x 10-3 M NaOH solution. c) A solution that contains

We are faced with different types of solutions that we should know how to calculate the pH or pOH for. These include calculation of pH for 1. Strong acids. - ppt video

Calculate the pH of a 1 x 10 ^-8 M HCl solution 1 X 108M solution of IJCI - Chemistry - Hydrogen - 13435683 | Meritnation.com

SOLVED: What is the pH of a 2.0 x 10^-7 M HCl solution? Hint: consider the background H3O+. pH = 6.61 pH = 6.70 pH = 6.79 pH = 6.52
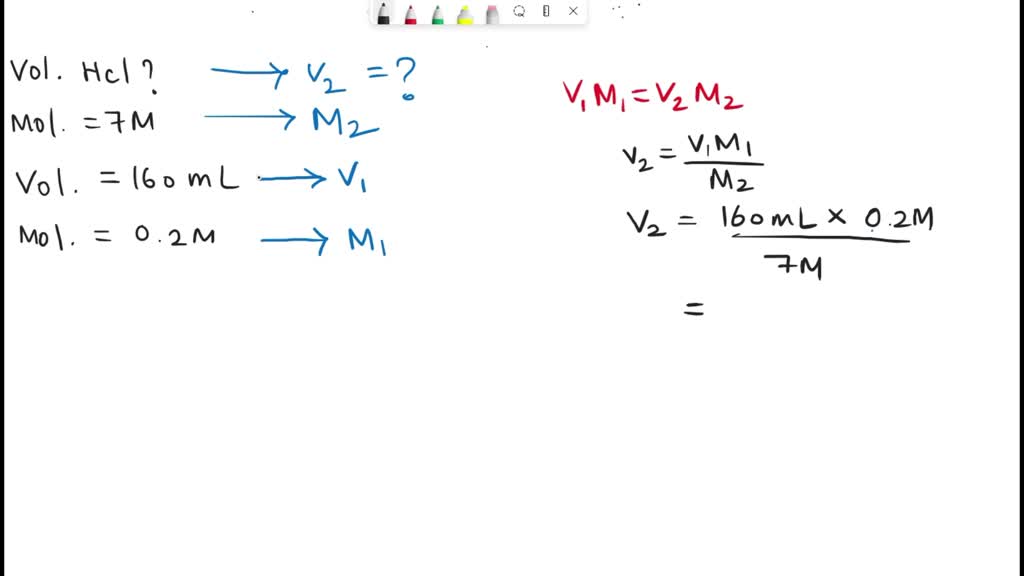
SOLVED: 2. How many ml of 7 M HCL solution is required to prepare 150 mL of 0.2 M HCL solution? Calculate the pH of the resulting dilute acid solution.



![Solved Calculate the pH of each solution. a) [H30+] = 1.7 x | Chegg.com Solved Calculate the pH of each solution. a) [H30+] = 1.7 x | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F0fa%2F0fa98cc3-4673-4562-9448-02d65a91573c%2Fimage)

![Malayalam] The pH of a 10^(-7) M aq. solution of HCl at 298 K is 7 Malayalam] The pH of a 10^(-7) M aq. solution of HCl at 298 K is 7](https://static.doubtnut.com/ss/web/9899943.webp)


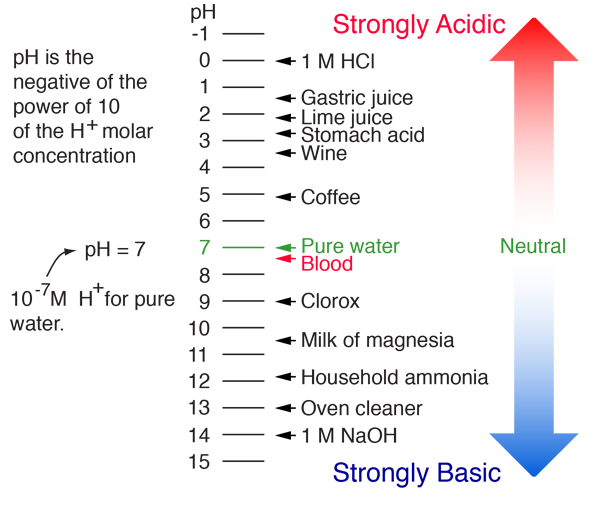
:max_bytes(150000):strip_icc()/how-to-calculate-ph-quick-review-606089_final-165915b0177b4f6e82843f25097f51df.png)